TIJ-BK119, the most recent addition to Domino Printing Sciences’ (Domino) array of specialized coding and marking solutions, is fast-drying, offers high contrast, and is appropriate for application on medical devices and personal protective equipment (PPE). This product contributes to the ongoing efforts against COVID-19.
Chat With A Coding, Marking & Labelling Expert Today
The ongoing pandemic has led to various adaptations in manufacturing technology and supplies, and BK119 stands as another illustration of this trend. Originally designed for applications in the food industry, this newly introduced ink for Domino’s Gx-Series thermal inkjet (TIJ) printers, deemed safe for food packaging, complies with EuPIA’s Good Manufacturing Practice protocols. Notably, it is now being employed by manufacturers producing particle-filtering respirator face masks intended to safeguard frontline workers during the COVID-19 crisis.
James Gibbins, Product Manager – Fluids at Domino, notes, “Respirator face masks are classified as personal protective equipment, while surgical masks fall under the category of medical devices – both are subject to compulsory coding requirements within the countries in which they are sold.” As the demand for these products continues to surge, Domino, drawing on its extensive experience in providing solutions to the medical device industry, remains committed to enhancing its products, consumables, and services to meet evolving coding requirements for PPE.
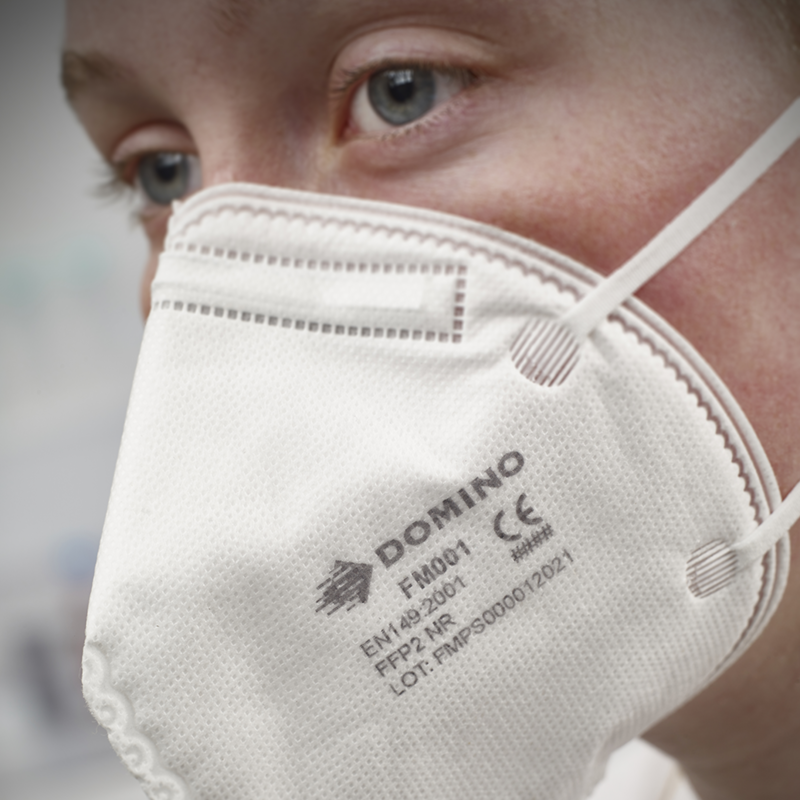
In contrast to surgical masks, which aim to limit the dispersal of saliva droplets to protect others and the external environment, respirator face masks are specifically designed to safeguard both the wearer and others. They achieve this by filtering out harmful substances and viruses in both inhalation and exhalation. Thermal inkjet (TIJ) is typically the preferred coding method for both respirator and surgical masks, as maintaining legibility and contrast is crucial for compliance with regulatory standards like the European Union Regulation 2016/425 on Personal Protective Equipment and Regulation 2017/745 on Medical Devices.
Respirator face masks adhere to ISO and EN standards, requiring a ‘CE logo’ and details about the filter type (e.g., FFP1, FFP2, and FFP3 in Europe), which varies by the region of sale. BK119, among TIJ inks, offers exceptionally high contrast for optimal legibility, making it suitable for all regulatory coding types, including Data Matrix and 2D codes. James Gibbins, Product Manager – Fluids at Domino, emphasizes, “BK119 was developed to ensure maximum code quality and legibility. In addition to meeting compliance requirements of manufacturers of medical devices and PPE, it is also fast drying with excellent adhesion properties, making it an ideal coding solution for non-porous respirator face masks.”
Respirator masks are typically manufactured on high-speed production lines, with coding applied before the masks undergo die-cutting to achieve their final form. Throughout this process, masks come into contact with various machinery, both during die-cutting and subsequent packaging. To minimize the risk of codes becoming smudged or unreadable, a durable and fast-drying ink is essential.
Comprising four non-porous layers, including a polypropylene filter and acrylic backing plate, respirator face masks may include moisture barriers depending on the filtration level for added protection against airborne contagions. Since respirator masks, particularly those with non-porous materials, do not readily absorb water-based ink, the ethanol-based BK119 proves to be a highly suitable solution. For other mask types, such as porous surgical masks, Domino offers water-based food packaging inks like BK652 and BK651 as appropriate alternatives.
Volker Watzke, EU Medical Devices Sector Manager at Domino, affirms, “BK119 highlights Domino’s ongoing commitment to helping manufacturers through COVID-19 and beyond. Throughout the pandemic, manufacturers have been faced with the need to adapt to the current circumstances, and many have stepped up to produce medical equipment for the first time. We are pleased to be able to support the supply of significant volumes of face masks, COVID-19 test kits, and other PPE globally by rapidly developing and adapting both products and consumables to meet the demands of our customers. Our sector-related project management team works to meet the specific needs of individual customers in the medical and PPE sectors, with rapid sampling and implementation processes to meet evolving needs while adhering to industry standards.”
To date, Domino has delivered over 1,000 TIJ printheads worldwide for face mask applications, including more than 400 printheads to a single factory in China. However, TIJ is not the sole technology contributing to the fight against COVID-19. Domino has also provided hundreds of V-Series thermal transfer overprinters and Ax-Series continuous inkjet printers for coding on the external packaging of surgical face masks. Furthermore, there has been a notable surge in demand for V-Series printers and D-Series high-speed laser coders for COVID-19 diagnostic test kit packaging applications. Since January 1st, over 40 D-Series laser coders have been supplied for these purposes in Korea alone.
Another swiftly expanding area for Domino is in the production and distribution of vaccines. As vaccine manufacturers strive to enhance capacity, Domino is expediting equipment delivery worldwide. A prominent Chinese vaccine producer urgently ordered Domino M-Series labellers for coding vaccine packaging. Domino’s manufacturing and service facilities in Sweden and China collaborated to fulfill the order, and the machines, representing several months of demand, were air-freighted and installed in under three weeks. Given the critical nature of vaccines, Domino is collaborating with customers to integrate vision solutions for code verification and to assess the integrity of individual vials and syringes. As a global player with a leading role in the life sciences industry and a comprehensive coding and marking solution portfolio, Domino remains committed to aiding customers worldwide in the ongoing global fight.
Jill Woods, Product Compliance Officer at Domino, reflects, “The last 12 months have brought about profound change on a global level, with manufacturers from across industries coming together to address public health needs. As a member of EuPIA, Domino’s manufactured inks – compliant to GMP – have enabled manufacturers to comply with the legal coding requirements for particle-filtering respirator face masks, as well as surgical masks, and additional PPE. We are committed to helping our customers to navigate the challenges posed by the global pandemic. We will continue to demonstrate our expertise in formulating reliable, safe solutions in our advanced fluids manufacturing facilities in the UK and China, and work to ensure a steady supply of goods and services via our vast worldwide distribution network.” For more information on Domino’s solutions for medical devices, PPE, and medical-grade face masks, please reach out.
[1] PR Newswire, ‘Recent Uptick in COVID-19 Outbreak is Driving Growth of Surgical Masks Demand’, https://www.prnewswire.co.uk/news-releases/recent-uptick-in-covid-19-outbreak-is-driving-growth-of-surgical-mask-demand-872302624.html

